Moreover, the molecule's bond order, which refers to the number of bonds between a pair of atoms, is #"1/2"#, another aspect that supports the hybrid model. However, like I've said, experimental data would point towards the hybrid model partial charges on all atoms would be present, and the molecule's double bonds have single bond character as well. So, in theory, this structure would be more stable than the previous two based on the fact that it has more covalent bonds and no formal charges an any of the atoms. Since sulfur has access to its 3d-orbitals, it's perfectly capable of expanding its octet to accomodate 10 electrons instead of 8. This time no formal charges are present - each oxygen atom needs 6 electrons and gets 6 electrons, the same being true for sulfur. The charges on the atoms are #"+1.4"# for sulfur and #"-0.7"# for each oxygen atom.Īnother Lewis structure that can be drawn for #SO_2# is this one The negative charge will be split on the two oxygen atoms. In this case, the actual structure would be a hybrid that would look like this Both structures have formal charges - the negative formal charge is placed on the more electronegative atom - oxygen, while the positive charge is placed on sulfur, the less electronegative of the two atoms. These two resonance structures are equivalent and will contribute equally to the hybrid structure. A couple of important things to notice here.  
The first two Lewis structures that can be drawn for #SO_2# will beĪll 18 valence electrons are accounted for - 6 electrons from 3 bonds and 10 electrons distributed as lone pairs on the three atoms. The total number of valence electrons we have at our disposal is 18 - 6 from sulfur and 6 from each of the two oxygen atoms. Let's draw the first two Lewis structures for #SO_2#. However, a third Lewis structure can be drawn for #SO_2# which is more stable in theory, but doesn't quite match experimental data. The third structure is identical to the second with respect to formal charges, but this time the oxygen on the left will get a (-1) formal charge and the one on the right a (+1) formal charge.Sulfur dioxide, or #SO_2#, has two resonance structures which contribute equally to the overall hybrid structure of the molecule. 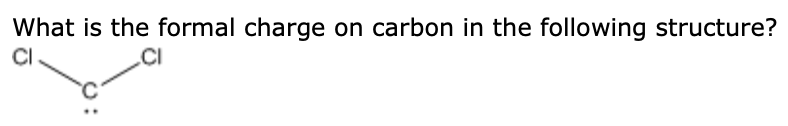
The oxygen on the right forms 1 bond with the carbon and has 3 lone pairs, for a total of 7 electrons since it has one more electron than it needs, it will automatically have a (-1) formal charge. This will result in a (+1) formal charge. This means that it will get 5 electrons - 3 from the bonds and 2 from the lone pair now it has one less electron than it "needs", i.e. Notice the oxygen on the left now forms 3 bonds with the carbon and has 1 lone pair instead of 2. However, things have changed for the oxygen atoms. Carbon is in the same position it was earlier - it forms 4 bonds #-># zero formal charge. Since oxygen has 6 valence electrons, it will have a zero formal charge. In addition to these electrons, they both have 2 lone pairs this brings the total number of electrons an oxygen atom gets to 6 (2 + 4). Both of them form 2 bonds, which means they get 2 electrons. Since carbon has 4 valence electrons, its formal charge will be zero. Carbon forms 4 bonds, which means it gets 4 electrons - 1 from each bond. Let's start with the first Lewis structure. The easiest way to assign a formal charge on an atom is to compare the number of valence electrons that atom has with how many electrons it "gets" in a molecule - assuming bond electrons are shared equally at all times regardless of electronegativity. The carbon dioxide molecule has a total of 16 valence electrons - 4 from the carbon atom and 6 from each of the two oxygen atoms, all of which being accounted for in the three Lewis structures above. SIDE NOTE: the actual structure of the carbon dioxide molecule is a hybrid between these three structures, but I'll just show you each of them separate because I don't want the answer to become too long. In order to determine formal charges for the atoms in the carbon dioxide molecule you need to take into account the fact that #"CO"_2# has three resonance structures that look like this: 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
